At a hydrophobic interface the interactions are anisotropic directional because the hydrophobic substance does not form hydrogen bonds. One contains water and acts as a reference cell the other contains the sample.
Free Energy Endergonic Vs Exergonic Reactions Article Khan Academy

16 4 Free Energy General College Chemistry Ii

Gibbs Free Energy Change Dg And Entropy Change Ds Secondary Science 4 All
The microcalorimeter needs to keep these two cells at exactly the same temperature during the course of an experiment.
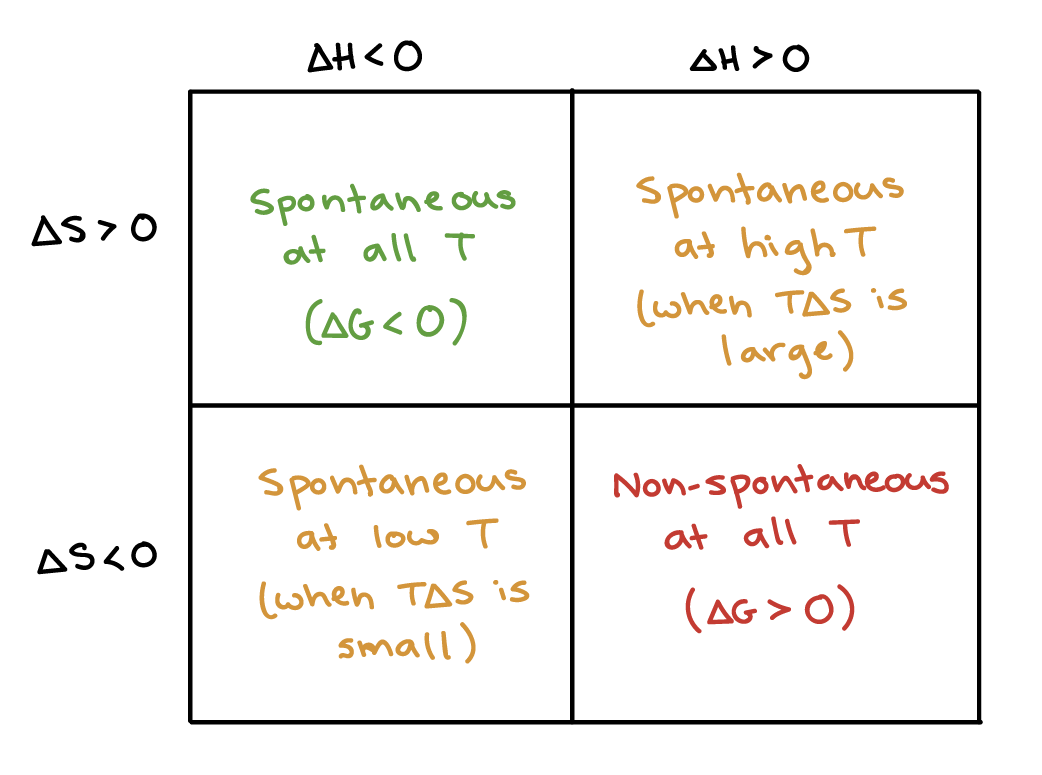
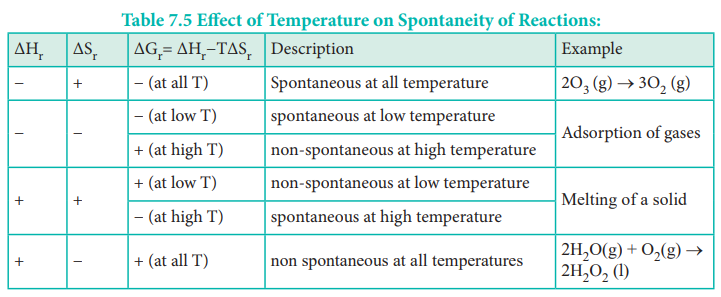
δg δh tδs. If ΔG 0 then the system is at the limit of reaction spontaneity. Therefore the reaction becomes spontaneous when T 469 K 196 ºC. ΔG reaction ΔH reaction - TΔS reaction.
At higher temperatures -TΔS will become more and more negative and will eventually outweigh the effect of ΔH. 441 calculate equilibrium concentrations given suitable data. In the equation ΔG ΔH - TΔS.
In an ITC experiment ΔH of binding is measured directly. ΔH es positivo lo que indica que la reacción es endotérmica. ΔGf Cu2S s -862 kJmol.
So the mixing of oil and water has a positive ΔS. Monomer List in csv. ΔG ΔH TΔS.
The entropy of the system is increased due to the mixing of hydrophilic molecules with water and then the change in entropy ΔS is a positive value. ΔG system ΔH system - TΔS system And therefore we have derived from the 2nd Law of Thermodynamics. ΔG ΔH TΔS.
Het List in SDF. Gibbs had devised a method of predicting ifwhen a process is spontaneous based upon thermodynamic properties of the. Reaction is at equilibrium.
This entropic effect leads to an unfavorable free energy of mixing oil and water ΔGΔH-TΔS 0. ΔG -93000 - T x -198 note that the enthalpy is given in kilojoules. So the spontaneity of solution formation whether or not a solution will form depends on the sign of the.
Note that to be useful as a fuel acetylene must release lots of energy spontaneously and a release of 12996 kJ per mole of acetylene consumed sounds quite a sensible amount when compared to other hydrocarbon fuels. When ΔH is negative and ΔS is positive the sign of ΔG will always be negative and the reaction will be spontaneous at all temperatures. Δg Δh tΔs ii Self Dimer.
Como tanto ΔH como TΔS son positivos el signo de la energía libre ΔG ΔH - TΔS dependerá del valor de T. ΔHfSO2 g -297 kJmol SmSO2 g 248 JK mol Then determine ΔGrxn of the following reaction. In the dissolution process steps 1 and 2 listed above require energy because.
The reaction is spontaneous and exergonic. Then the change in enthalpy is a negative value. ΔG soln ΔH soln TΔS soln 1 solution formation is spontaneous occurs when ΔG soln 0 ΔG soln is negative.
Entropy always increases with solution formation in other words ΔS soln 0 ΔS soln is always positive. Ii If ΔG is positive. This equation is called the Gibbs Helmholtz equation.
For this ideal case the fuel energy is converted to electrical energy at an efficiency of 23712858 x100 83. The ITC200 microcalorimeter has two cells. ΔG reaction 0.
Determine ΔGf for SO2g. The free energy G describes both the energetics ie the enthalpy H and the randomness or probability ie the entropy S of a process ΔGΔH-TΔS where T is the absolute temperature. ΔG ΔH -TΔS -1 which is the same as that calculated in the question.
1在温度压力一定的条件下放热的熵增加的反应一定能自发进行 2在温度压力一定的条件下吸热的熵减少的反应一定不能自发进行 3当焓变和熵变的作用相反时如果二者大小相差悬殊可能某一因素占主导. This is far greater than the ideal efficiency of a generating facility which burned the hydrogen and. 从公式 ΔgΔhtΔs 看化学反应的推动力Δg依赖于两个量①形成或断开化学键所产生的能量变化 Δh② 体系无序性变化与温度的乘积这个公式后来被称为吉布斯方程它是化学中最有用的方程之一.
434 deduce from a concentration-time or a rate-concentration graph the rate of reaction andor the order with respect to a reactant. 425 use the equation ΔG ΔH - TΔS to calculate standard free energy changes. By combining these two expressions we arrive at an expression describing the system at equilibrium at constant pressure.
TΔS is positive and so -TΔS is negative. Since new bonds are formed between hydrophiles and water molecules this mixing is exothermic. The enthalpy and entropy changes that occur in the dissolution process are shown in Figure 2 below.
ΔG ΔH - TΔS -28583 kJ 487 kJ -2371 kJ. The equation becomes ln k p ΔH 0 RT ΔS 0 R. A primer self-dimer is formed by intermolecular interactions between the two same sense primers where the primer is homologous to itself.
ΔG ΔH - TΔS ΔG -1125 kJmol - 298K -01314 kJmol K -7234 kJmol 20. According to the second law of thermodynamics entropy of the universe always increases for a spontaneous process. The integration constant can be calculated from the thermodynamic entropy relation ΔG 0 ΔH 0 TΔS 0.
The above relation is the differential form of the Vant Hoff equation but the greater the value of ΔH 0 the faster the equilibrium constant changes with temperature. Assume 25 C for all reactions. ΔG and ΔH are protected under issued patents and pending patent applications.
Se pasa de una molécula en estado sólido a una molécula de sólido y otra de gas con lo que el desorden aumentará ΔS positivo. ΔG ΔH -TΔS. ΔG ΔH-TΔS pH Enzymatic Assay pH ITC Substrate or Competitor.
At constant temperature and pressure the Gibbs equation is ΔG ΔH - TΔS in which ΔH is the change in enthalpy ΔS is the change in entropy and ΔG is the amount of free or available energy. ΔG ΔH - TΔS. 43 Rates of reaction.
Gibbs free energy is a derived quantity that blends together the two great driving forces in chemical and physical processes namely enthalpy change and entropy change. We are therefore. ΔGΔHTΔS 体の等温変化であるから ΔH系 0 ΔG系 0 300 K 0 の氷の融解熱は602 kJmol1また100 での水の蒸発熱は407 kJmol1である0 氷が 解 圧変化では q可逆 ΔHであるから 1 1 3 1 22 1 J K mol 273 K 6 02 10 J mol 融 融.
The reaction is non-spontaneous and endergonic. And the condition for the chemical reaction or physical change to be at equilibrium that is. And T 93000198 Kelvin.
Number of Data Targets. US 8642654 US 10051880 CA 2734720C US 16038513 US 15914676 CA 2527940C US 9211275 US 10154982 US 16203748 US 16881597 US 10660958 CA 2790790C US 14390495 CA 2873092C US 14453999 CA 2919778 US 14213713 CA 2905225 US 11287803. When ΔG 0 then T x -198 -93000.
As for the results. Why does Gibbs free energy have to be negative. Special tools 3D Structure Series.
For a spontaneous process ΔH system - TΔS system 0 For a spontaneous process ΔG system 0 And there you have it. ΔG ΔH TΔS Gibbs energy change enthalpy change temperature x entropy change ΔG gives a criteria for spontaneity at constant pressure and temperature i If ΔG is negative 0 the process is spontaneous. Now increasing the temperature will change things.
Gibbs Free Energy G

2020 A Level H2 Chem P1 Q11
19 6 Free Energy And Temperature Chemistry Libretexts

Gibbs Energy
Gibbs Free Energy Chart

Gibb S Energy Its Importance And Its Relation With Spontaneity Of A Reaction
How Is Gibbs Free Energy Related To Enthalpy And Entropy Socratic
The Gibbs Free Energy Post 16 Thermodynamics Tutorials Resource Rsc Education
